|
6/24/2023 0 Comments As element charge is
We can calculate the effective nuclear charge by subtracting the number of inner shell electrons from the number of protons. How to Calculate Effective Nuclear Charge Because of the varying charge on electrons in different orbitals, we typically refer to the effective nuclear charge, which is the effect of the nucleus experienced by the outermost electron of the atom, taking into account the shielding effect of inner electrons. 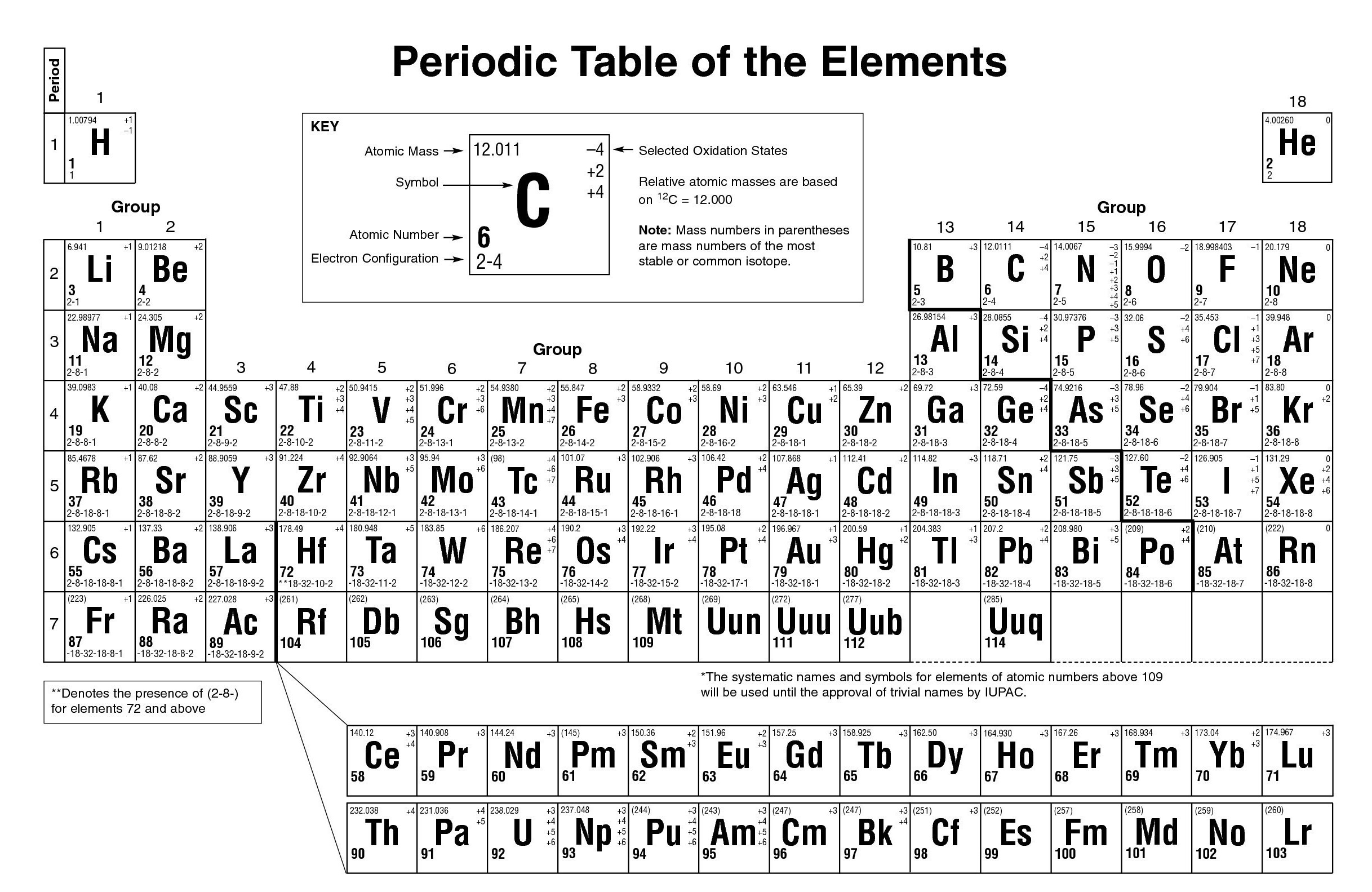
The presence of electrons on the inner shells of an atom “shield” the outermost electron from feeling the full positive charge. This results in a varying attraction of the nucleus on the electrons surrounding the nucleus, which is known as nuclear charge. However, negatively charged electrons around the nucleus are organized into layers called orbitals which repel each other, and negate some of the positive charge of the nucleus. The formal charge is the 'charge an element would have in a molecule or ion if all of the bonding electrons were shared equally between atoms HR: N Based on the Lewis structure given the formal charge on the central nitrogen atom is F: The formal charge is the charge an. Electrons are attracted to the nucleus as they are negatively charged. Solution Formula of formal charge V - N - (B/2). The nucleus of an atom contains positively charged particles called protons. The net attraction on these outer electrons is known as effective nuclear charge. Group 2 elements are alkaline Earth Metals with a. Electrons are attracted to the nucleus as it is positively charged, but electrons in the inner shells can negate some of the attraction of the nucleus on the outermost electrons. Group 1 elements are the alkali metals that have a charge of 1. Nuclear charge is a measure of the ability of protons in the nucleus to attract the negative electrons in orbit around the nucleus. The equation for calculating nuclear charge is Zeff = Z - S, where Zeff is the effective nuclear charge, Z is the number of protons, and S is the number of inner electrons.ģ. If an element is bonded to 4 other atoms and has a formal charge of 1, what group must the element be in a. Metals lose electrons to form positively charged ions and nonmetallic elements gain electrons to. 
A certain element forms an ion with 54 electrons and a charge of 2. Atoms gain or lose valence electrons to become more stable. You can calculate effective nuclear charge if you know the number of inner electrons and the number of protons of an atom, both which can be found either from the periodic table or from online resources. A certain element forms an ion with 10 electrons and a charge of 2. How do you calculate effective nuclear charge? Nuclear charge values have been determined for the elements. These values are recorded in encyclopedias, scientific textbooks, and scientific journal articles.Ģ. How do you find effective nuclear charge? The circular journey ticket will be charged for two single journeys, each journey being taken as half of the total distance. 
These outer electrons are also known as valence electrons.ġ. Elements in different groups on the periodic table have different numbers of electrons in their outermost shells. Group one elements have a 1 charge, while elements. These negatively charged electrons are arranged into shells which form layers surrounding the nucleus. The charge of elements in the periodic table can be determined by looking at the groups they belong to. So there's no simple rule for learning the number of electrons an atom may gain or lose in a compound (and using electricity, for example, it's possible to remove electrons well below the valence shell), though the number of electrons in the valence shell is unique.Refresher: Atoms are composed of a nucleus, containing positively charged protons and neutral neutrons, surrounded by a cloud of negatively charged electrons. " So an element has a specific valence, depending on its group (e.g., C, 4 or Xe, 0), but may have multiple values for valency, such as in $\ce$. It's not simply the column that determines the ionic charge of an element, but also other factors, such as row and with what other elements it's combined.Įpediaa states, "valence refers to the ability of an atom to be combined with another atom whereas valency refers to the maximum number of electrons that an atom can lose or gain in order to stabilize itself.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
